 
ISO 13485 is an effective solution to meet the global requirements for a quality management system (QMS). Iso 13485 & 21 Cfr 820 Template Documentation Operational Procedure Qop 42 01 Control of Documents - Download as PDF File (.pdf), Text File (.txt) or read online. Control of Documents (4.2.3) Control of Records (4.2.4). 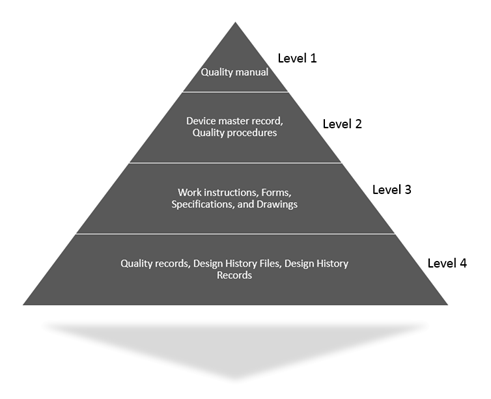
ISO 13485 Store > ISO Requirements > ISO 13485 Documentation Requirements. 
› ▆ Iso 13485 Sop Documents ▆ĭ126: DEMO OF ISO Medical Device Document Kit Price 399 USD Total editable documentation package Complete editable document.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
